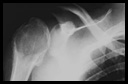
 History: A 19 year-old male with an increasingly painful right arm was found by x-ray to have a circumscribed lytic lesion located in the proximal humeral epiphysis (Fig. 1). A radiograph demonstrated irregular focal thinning of the cortex as well as a mild joint effusion. A subsequent curettage specimen produced an aggregate of red-tan soft tissue admixed with bone. Microscopically, a synovial lining was undermined by a solid, hypercellular tumor proliferation of round to polygonal cells (Fig. 2). The cells were of intermediate size with well-defined cytoplasmic borders, pink to focally clear cytoplasm and folded or clefted nuclei. Many multinucleated osteoclast-like giant cells were present as were numerous foci of immature bluish-pink cartilage-like material (Fig. 3). There were also calcifications around individual tumor cells resulting in a reticular chicken-wire pattern. Mitotic activity was low. The neoplastic cells were S-100 positive (Fig. 4).
History: A 19 year-old male with an increasingly painful right arm was found by x-ray to have a circumscribed lytic lesion located in the proximal humeral epiphysis (Fig. 1). A radiograph demonstrated irregular focal thinning of the cortex as well as a mild joint effusion. A subsequent curettage specimen produced an aggregate of red-tan soft tissue admixed with bone. Microscopically, a synovial lining was undermined by a solid, hypercellular tumor proliferation of round to polygonal cells (Fig. 2). The cells were of intermediate size with well-defined cytoplasmic borders, pink to focally clear cytoplasm and folded or clefted nuclei. Many multinucleated osteoclast-like giant cells were present as were numerous foci of immature bluish-pink cartilage-like material (Fig. 3). There were also calcifications around individual tumor cells resulting in a reticular chicken-wire pattern. Mitotic activity was low. The neoplastic cells were S-100 positive (Fig. 4).
Diagnosis: “Chondroblastoma, Proximal Humerusâ€
Yvonne Noronha MD, Donald R Chase, MD
Department of Pathology and Human Anatomy, Loma Linda University and Medical Center, Loma Linda, CA
California Tumor Tissue Registry, Loma Linda, CA
Discussion: Chondroblastoma (CB) is a rare osseous neoplasm closely related to giant cell tumor (GCT). Like GCT it has a marked predilection for the epiphysis and contains numerous osteoclast-like giant cells. It differs, however in that there are round to polygonal chondroblasts which form a cartilaginous matrix. CBs are 1/10th as common as GCTs and account for < 1% of primary bone tumors. Males are more commonly affected (2:1). Ninety percent of CBs occur before the age of 20 and mostly involve long bones particularly the proximal humerus and knee (70%). They also occur in flat bones and pelvic bones. Clinical presentation is usually chronic pain. The radiographs generally show a lytic lesion sometimes with stippled calcifications and possibly a joint effusion. In long bones CBs almost invariably involve the epiphysis but may also directly extend into the metaphysis and sometimes even into the diaphysis. The curetted material is described as being lobular, soft and friable, gray-pink and gritty. The effusion fluid is generally rust or straw-colored. Microscopically, the neoplastic cells are fairly uniform polygonal cells which stain for S100 protein. They contain lightly-staining or clear cytoplasm with sharply defined membrane borders. The nuclei are round or ovoid and commonly show clefts, grooves or invaginations. Mitotic figures can be present but atypical mitoses are never seen. Curiously, 20-30% of CBs have mildly atypical cells with enlarged, irregular, hyperchromatic nuclei. Their presence, however, does not indicate an adverse prognosis. Multinucleate osteoclast-like giant cells which stain for histiocytic markers (i.e. CD68) are common. Of considerable diagnostic importance are the areas of chondroid differentiation which may be a dominant feature or be scant and/or widely distributed. Cells in these diagnostic regions are relatively immature, lacking lacunae of normal cartilage. The accompanying matrix, unlike mature cartilage, may be eosinophilic. Calcifications may be seen within the matrix or more commonly around individual cells in what is described as a “chicken-wire†pattern. These calcifications†typical of chondroblastoma are not seen in its mimics. Secondary changes of aneurysmal bone cyst formation are found in 20-30% of the cases. Other cystic changes may include formation of unilocular or multilocular spaces filled with serous fluid. Occasional CBs may also show focal regions of necrosis. The differential diagnosis of chondroblastoma includes: • Giant cell tumor. Distinction from CB is important because GCT carries a 50% recurrence rate while CB recurs around 10%. GCTs are usually larger, radiographically more “aggressive†and are usually seen in skeletally mature individuals. Like CB, the neoplastic cell is the mononuclear stromal cell which bundles together to form the giant cells. Unlike CB, neither a chondroid matrix nor chicken wire calcifications are seen, and differing with CB, the stromal cells of GCT are negative for S100. • Eosinophilic granuloma. The neoplastic cell, labeled “Langerhan’s cellâ€, is cytologically different from those of a CB (or GCT). They have abundant pink cytoplasm and irregular grooved nuclei. The Langerhan’s cell is both S100 and CD1a positive and ultrastructure shows the presence of tennis racket-appearing “Birbeck granulesâ€. The chondroid matrix and chicken wire calcifications of CB are absent. • Chondromyxoid fibroma (CMF) is another benign neoplasm of undifferentiated cartilage composed of lobules of stellate cells in an abundant myxochondroid matrix separated by cellular regions of spindled to round cells and multinucleated giant cells. Like chondroblastoma, CMF commonly occurs in young males, but unlike CB it arises in the metaphysis and lacks significant calcification. • Clear cell chondrosarcoma is a most uncommon tumor which usually involves the epiphysis and may contain a large number of giant cells. Focally it may contain cells indistinguishable from chondroblasts, however unlike CB it is usually seen in older patients (>40 yrs) and shows broad sheets of cytologically atypical cells.
Chondroblastoma is a benign neoplasm, which, because of its low recurrence rate, can be successfully treated with curettage and bone chip grafting. An “en-bloc†resection is rarely needed. The recurrence rate is about 10%, usually within 3 years. The recurrent tumor is likely to be cured by either a second curettage or resection.
Despite the uniform benign, indolent nature of usual chondroblastomas, a variant occurs which has been given the name “aggressive chondroblastoma†(ACB). This variant is larger and more destructive than usual CB. Like a GCT, ACB may erode through the cortex and invade soft tissue. This behavior is relatively more common in pelvic lesions. Since many ACBs are microscopically indistinguishable from conventional chondroblastoma, radiographic interpretation is vital. However, in some cases focal pleomorphism may resemble that of a pleomorphic sarcoma, implying that a form of dedifferentiation from a pre-extent CB has taken place. Pulmonary metastases rarely may be seen even up to 30 years after the initial diagnosis. Despite these extraordinary examples, like usual CB, ACB typically is non-progressive and successfully treated with surgery.
Suggested Reading:
de Silva MV; Reid R. Chondroblastoma: varied histologic appearance, potential diagnostic pitfalls, and clinicopathologic features associated with local recurrence. Ann Diagn Pathol 7(4):205-13, 2003.
Lin PP; Thenappan A; Deavers MT; Lewis VO; Yasko AW. Treatment and prognosis of chondroblastoma. Clin Orthop Relat Res 438:103-9, 2005.
Suneja R; Grimer RJ; Belthur M; Jeys L; Carter SR; Tillman RM; Davies AM. Chondroblastoma of bone: long-term results and functional outcome after intralesional curettage. J Bone Joint Surg Br 87(7):974-8, 2005.
Kilpatrick SE; Pike EJ; Geisinger KR; Ward WG. Chondroblastoma of bone: use of fine-needle aspiration biopsy and potential diagnostic pitfalls. Diagn Cytopathol 16(1):65-71, 1997.
Harish K; Janaki MG; Alva NK. “Primary” aggressive chondroblastoma of the humerus: a case report. Musculoskelet Disord 17(5):9, 2004.
Papachristou DJ; Goodman MA; Cieply K; Hunt JL; Rao UN. Comparison of allelic losses in chondroblastoma and primary chondrosarcoma of bone and correlation with fluorescence in situ hybridization analysis. Hum Pathol 37(7):890-8, 2006.