 History: Nine years after being diagnosed and successfully treated for an intracystic papillary carcinoma of the left breast, a 65 year old woman presented with a large firm mass in the contralateral (right) breast. A radical mastectomy with axillary lymph node dissection was performed.
History: Nine years after being diagnosed and successfully treated for an intracystic papillary carcinoma of the left breast, a 65 year old woman presented with a large firm mass in the contralateral (right) breast. A radical mastectomy with axillary lymph node dissection was performed.
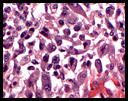
The excised tumor was 14 x 12 x 10 cm and well-circumscribed. It contained a central cavity lined by firm, white-tan partly gelatinous tissue and filled with brown-tan fluid admixed with necrotic debris. Microscopically, the viable portion of the tumor had alternating hypercellular and hypocellular areas as well as focal areas of coagulative necrosis (Fig. 1). Individual tumor cells varied greatly (Fig. 2) and included:
• small spindle shaped cells
• larger strap-like cells with abundant pink eosinophilic cytoplasm
• rounded cells with abundant pink cytoplasm and eccentric nuclei, and
• bizarre pleomorphic cells (Fig. 3) with occasional multinucleated tumor giant cells.
The axillary lymph nodes were benign.
Immunohistochemistry showed the cells to strongly decorate for vimentin (Fig. 4). Stains for S100, HMB45, cytokeratin, EMA, estrogen and progesterone receptors were negative. Myogenous markers for myogenin, desmin, muscle specific actin and smooth muscle actin also failed to stain.
Diagnosis: “Pleomorphic Sarcoma Mimicking Rhabdomyosarcoma, Breastâ€
Yvonne Noronha MD, Resa L Chase MD, Donald R Chase, MD
Department of Pathology and Human Anatomy, Loma Linda University and Medical Center, Loma Linda, CA,
Jerry L. Pettis VA Medical Center, Loma Linda, CA,
California Tumor Tissue Registry, Loma Linda, CA
Discussion: Primary breast sarcomas are extremely rare neoplasms accounting for less than 0.1% of all malignant tumors of the breast. In a series by Adem et al, only 25 cases of primary breast sarcomas were found in the Mayo Clinic files from 1910 to 2000. They comprised <0.0006% of their mammary malignancies. Due to the extreme rarity of these tumors the diagnosis of pleomorphic sarcoma of the breast is one of exclusion and includes the use of immunostains to exclude more common entities such as: • Spindle cell (sarcomatoid)/metaplastic carcinoma • Malignant melanoma • Malignant phyllodes tumor, pleomorphic variant • Pleomorphic liposarcoma • Pleomorphic rhabdomyosarcoma (so-called “adult†form) Spindle cell (sarcomatoid)/metaplastic carcinoma is recognized by the presence of an epithelial morphology and/or keratin/epithelial immunopositivity in the neoplastic spindle cells. Malignant melanoma may assume many phenotypes including those of pleomorphic sarcoma. Melanocytic markers such as Melan A, S100 and HMB45 may be helpful. Malignant phyllodes tumor may assume a pleomorphic appearance but usually maintains a biphasic pattern of spindled cells as well as pleomorphic cells. The leaf-like architecture usually remains and sometimes can be found only in the study of multiple sections. Pleomorphic liposarcoma contains areas of clearly identifiable vacuolated cells with cytoplasmic lipid which indents the nucleus. These lipoblasts help separate the tumor from other pleomorphic tumors which may appear identical. S100 may be positive in some of the tumor cells, but is of little diagnostic value. A diagnosis of pleomorphic rhabdomyosarcoma should not be made unless there is incontrovertible evidence of striated muscle differentiation in the form of cross striations or the demonstration of specific ultrastructural or immunohistochemical markers. The latter includes: • Myogenin which can be demonstrated in tumor cells of virtually all rhabdomyosarcomas. • Sarcomeric Actin as well as Common Muscle Actin which mark virtually all rhabdomyosarcomas. Little or no positivity is seen for Smooth Muscle Actin. • Desmin decorates tumors of both smooth and striated origin. It is usually positive in rhabdomyosarcomas particularly in rounded rhabdomyoblasts or strap cells. • Myosin may also be expressed in striated muscle neoplasms. • Myoglobin is only expressed in rhabdomyosarcomas with a high degree of differentiation. However, interpretative caution must be taken since myoglobin may also be released by adjacent injured skeletal muscle fibers with subsequent absorption by tumor cells. Our presentation case, despite extensive sampling, failed to show the morphology of an epithelial malignancy and was cytokeratin negative helping to exclude sarcomatoid/metaplastic, carcinoma. Although the tumor had large eosinophilic pleomorphic strap-like cells, no definite cross-striations were observed. Negativity for muscle markers helped in the exclusion of rhabdomyosarcoma, and negativity for S100, HMB45, estrogen and progesterone helped exclude melanoma and other tumors. The strong vimentin positivity in this setting helped confirm mesenchymal origin. During the past years, malignant fibrous histiocytoma (MFH) has been used as a kind of diagnostic waste-basket for hard to classify sarcomas such as our presentation case. The constituent cells of MFH have not yet been found to express antigens unique to the tumor and partly for this reason serious doubt has been raised about the existence of MFH as a specific entity. The term “pleomorphic sarcoma†is now gradually replacing “MFH†and through immunohistochemistry, molecular, genetic and other studies many tumors previously classified as MFH are being re-classified into more specific categories. The prognosis and treatment for primary breast sarcomas is similar to that of most soft tissue sarcomas at other sites. Important prognostic factors include histologic type, tumor size and tumor grade. As with most other sarcomas, primary treatment is wide local excision with consideration given for radiation to the tumor bed. Since the tumors tend to metastasize via blood vessels, axillary lymph node dissection need not be routinely performed. Suggested Reading:
Pandey M; Mathew A; Abraham EK; Rajan B. Primary sarcoma of the breast. J Surg Oncol 1;87(3):121-5, 2004.
Adem C; Reynolds C; Ingle JN; Nascimento AG. Primary breast sarcoma: clinicopathologic series from the Mayo Clinic and review of the literature. Br J Cancer 19;91(2):237-41, 2004.
Rajaram V; Hill DA; Doherty GM; Liapis H; Dehner LP. Pleomorphic rhabdomyosarcoma of the anterior abdominal wall following multimodality treatment for carcinoma of the rectum. Int J Surg Pathol 12(2):161-5, 2004.
Leibl S; Moinfar F. Mammary NOS-type sarcoma with CD10 expression: a rare entity with features of myoepithelial differentiation. Am J Surg Pathol 30(4):450-6, 2006.
Gogas J; Kouskos E; Markopoulos C; Mantas D; Antonopoulou Z; Kontzoglou K; Stamoulis J; Kyriakou V. Carcinosarcoma of the breast: report of two cases. Eur J Gynaecol Oncol;24(1):93-5, 2003.
Tos AP. Classification of pleomorphic sarcomas: where are we now? Histopathology 48(1):51-62, 2006.
Rampisela D; Donner LR. Pleomorphic sarcoma as a mimicker of sarcomatous carcinoma. Ultrastruct Pathol 28(4):209-12, 2004.
Nogueira M; Andre S; Mendonca E. Metaplastic carcinomas of the breast–fine needle aspiration (FNA) cytology findings. Cytopathology 9(5):291-300, 1998.
Silver SA; Tavassoli FA. Pleomorphic carcinoma of the breast: clinicopathological analysis of 26 cases of an unusual high-grade phenotype of ductal carcinoma. Histopathology 36(6):505-14, 2000.
Erlandson RA; Woodruff JM. Role of electron microscopy in the evaluation of soft tissue neoplasms, with emphasis on spindle cell and pleomorphic tumors. Hum Pathol 29(12):1372-81, 1998.