History:A 69 year-old woman from Laos presented with hemoptysis and a 9 cm mass in the right middle and lower lung lobes (Fig. 1). It obliterated the right middle lobe bronchus, obstructed the inferior pulmonary vena cava and extended to the chest wall. Ten years prior, the tumor was recognized by CT scan, but therapy had not been given. Metastatic workup at the current presentation failed to show a different site of origin.
By needle biopsy the tumor was solid and hypercellular (Fig. 2, 3) and had a faint fascicular growth pattern (Fig. 4). The cells were primitive consisting of polygonal, rounded and spindled forms with vesicular chromatin and nucleoli (Figs. 5, 6, 7). Occasional lymphoid infiltrates were seen. Neither necrosis nor significant numbers of mitoses were seen.
The tumor immunohistochemical profile was:
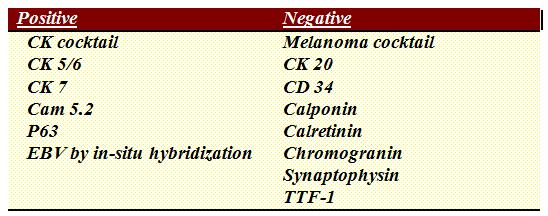
Molecular testing for EGFR mutation was negative as was a FISH study for ALK rearrangement.
Diagnosis: Primary Lymphoepithelioma-like Carcinoma of the Lung
Ying Zhao, MD, Anwar Raza, MD, Camilla Cobb, MD, Donald R Chase, MD
Department of Pathology and Human Anatomy,
Loma Linda University Medical Center, Loma Linda, California
California Tumor Tissue Registry, Loma Linda, California
Discussion: Pulmonary lymphoepithelioma-like carcinoma (LELC) is an undifferentiated squamous cell carcinoma associated with a prominent lymphocytic infiltration. This rare entity is defined as a subtype of large cell carcinoma of the lung according to the 2004 World Health Organization. LELC of the lung was first reported in 1987 (Begin et al.). It is mostly seen in Asians and only a few cases appear in the literature. Just 150 cases were reported prior to 2006, with nearly two-thirds of them arising in patients from Taiwan, Southern China and Hong Kong (Ho et al.).
Pulmonary LELC has a median occurrence age of 51 years, ranging from 9 years to 74. Gender is affected equally. Although 33% of patients are asymptomatic, most present with pulmonary symptoms (Liang et al.). Lung variants tend to be EBV positive, but those which arise in non-pulmonary sites such as stomach, salivary glands, thymus, skin, vagina, and urinary bladder tend to be EBV negative. Unlike other pulmonary carcinomas, there is no strong association with cigarette smoking.
Pulmonary LELC presents as a solitary, discrete nodule, usually sparing the major bronchi (Chan et al., Liang et al.). Most present at an early stage or are locally advanced. Microscopy shows sheets of primitive large cells with vesicular nuclei and prominent nucleoli, mixed with lymphocytes and plasma cells. The lymphoid infiltrate may be so prominent as to suggest lymphoma. Tumor cells usually express high levels of CK, CK5/6, and p63, indicating that LELC is epithelial in origin, and favored by Liang et al to be a variant of squamous carcinoma.
Histologically, pulmonary LELC is indistinguishable from the prototypical lymphoepithelial carcinoma of the nasopharynx. Consequently, nasopharyngeal origin needs to be excluded in all cases (Huang et al.). Imaging features suggesting primary pulmonary LELC include central location, large size, vascular encasement and peribronchovascular nodal spread.
The mainstay of treatment for early stage disease is surgical resection, whereas multimodality treatment (surgery, chemotherapy, and radiotherapy) has been adopted for advanced or metastatic disease (Ho et al.). In our case, the patient had a stage IV middle lobe large mass with massive extension which made the tumor unresectable and allowing for radiation and chemotherapy treatments. Subsequently, the patient had one cycle of cisplatin and VP-16 then was switched to xeloda due to intolerance and complications. The tumor grew slowly during the subsequent two years follow-up period.
LELC of the lung has a better prognosis than non-LELC (Han et al., Chen et al.). The significantly enhanced survival is particularly evident in stage II, III and IV. Tumor recurrence and necrosis (5% or more of tumor) are associated with a poor prognosis (Liang et al.). Of interest is the 10 year survival with the large mass under no medical treatment, seemingly supporting the concept that pulmonary LELC has a better prognosis then non-LELC.
In conclusion, LELC should be considered in the differential diagnosis of primary lung tumors, particularly when poorly differentiated spindled and polygonal cells with an extensive lymphocytic infiltrate are observed. Better prognosis and proper patient management require familiarity with this rare entity and accurate diagnosis.
Suggested Reading:
L. R. Begin et al, Epstein-Barr virus related lymphoepithelioma-like carcinoma of lung,
Journal of Surgical Oncology, vol. 36, no. 4, pp. 280–283, 1987.
Weiss et al. Analysis of lymphoepithelioma and lymphoepithelioma-like carcinomas of different organs for Epstein-Barr viral genomes by in situ hybridization. Am J Surg Pathol. 1989;13:625-631.
Han et al. Lymphoepithelioma-like carcinoma of the lung with a better prognosis. Am J Clin Pathol 2001;115:841-850.
Chen FF et al. Epstein-Barr virus-associated non-small cell lung carcinoma: undifferentiated “lymphoepithelioma-like†carcinoma as a distinct entity with better prognosis. Cancer 1998;82:2334-42.
Liang et al. Primary pulmonary lymphoepithelioma-like carcinoma; Fifty-two patients with long-term follow-up. Cancer, 2012 Oct 1;118(19):4748-58.
Chan JKC, Hui PK, Tsang WYW, et al. Primary lymphoepithelioma-like carcinoma of the lung. Cancer 1995;76:413-22.
Ooi GCet al. Computed tomography characteristics of advanced primary pulmonary lymphoepithelioma-like carcinoma. Eur Radiol 2003;13:522-6.
Ho JC, et al. Lymphoepithelioma-like carcinoma of the lung. Respirology 2006 Sep;11(5):539-545.
Ho JC et al. Capecitabine as salvage treatment for lymphoepithelioma-like carcinoma of lung. J Thorac Oncol 2009 Sep;4(9):1174-1177.
Huang et al. Lymphoepithelioma-like carcinoma of the lung: an unusual case and literature review. Case reports in pulmonology 2013 Oct: 143405-143408.