 History: A 57 year old male presented with a painless 5 x 3.5 x 2.5 cm mass in the superficial tissues of the right upper back. An excisional biopsy was performed.
History: A 57 year old male presented with a painless 5 x 3.5 x 2.5 cm mass in the superficial tissues of the right upper back. An excisional biopsy was performed.
Grossly, the mass appeared circumscribed and had a brown-tan lobulated appearance.
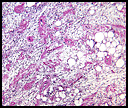
Microscopically, the mass consisted of an admixture of spindled cells and mature adipose tissue (Fig. 1). The spindled cells were arranged in alternating hypercellular regions and less cellular, partly myxoid regions (Fig. 2,3). Thickened bundles of collagen focally intersected the tumor. Numerous ectatic rounded blood vessels with prominent peri-vascular hyalinization were a striking feature, and were evenly distributed throughout the tumor (Fig. 3, 4). Neither mitoses nor significant cellular pleomorphism were seen and necrosis and hemorrhage were not encountered. The cellular areas of the tumor, showed a lesser amount of adipose tissue, and resembled classic solitary fibrous tumor (SFT). The less cellular, more myxoid areas, however, showed larger amounts of fat cells of varying sizes, and were more reminiscent of spindle cell lipoma (SCL). Curiously, there was an interface between the two phenotypes suggesting either a modulation between expressions of the same tumor or an abutment between two different, but morphologically similar processes (Fig. 4).
Immunohistochemistry showed the spindled cells to be strongly and diffusely positive for CD34, CD99, bcl-2 and vimentin, both in the cellular and the more myxoid areas (Fig. 5). Smooth muscle actin, muscle specific actin and desmin were negative. A stain for S100 protein marked the mature adipose tissue but not the spindled cells.
Diagnosis: “Lipomatous Variant of Solitary Fibrous Tumorâ€
Yvonne Noronha MD1, Resa L Chase MD1,2,3, Donald R Chase, MD1,3
1Department of Pathology and Human Anatomy, Loma Linda University
and Medical Center, Loma Linda, CA,
2Jerry L. Pettis VA Medical Center, Loma Linda, CA
3California Tumor Tissue Registry, Loma Linda, CA
Discussion: SFT was first described as a distinct entity by Klemperer and Rabin in 1931 as a pleural-based lesion which was thought to be mesothelial or sub-mesothelial in origin. Later the term “mesothelioma†was dropped in favor of the current designation of SFT. Morphologically identical tumors have been reported in a myriad of sites including liver, meninges, skin, respiratory tract, thyroid, orbit, peri-renal tissues and soft tissue. It is now felt that SFT is a mesenchymal neoplasm of fibroblastic and myofibroblastic origin. Useful features include:
• circumscription
• alternating hypercellular foci and hypocellular sclerotic foci
• bland, short spindly or ovoid cells with poorly defined cytoplasm and few mitotic figures (<4/10 HPF)
• haphazard, storiform or fascicular arrangement of spindle cells
• interwoven thin or thick collagen bundles separating the spindle cells
• CD34 positivity (essential)
• Vimentin and CD99 positivity with variable expression of bcl-2.
Spindle cell lipoma was originally described by Franz Enzinger in 1975 as a relatively small superficial fatty tumor in the neck, shoulder and upper back, typically in middle-aged men. It classically consists of mature fat, collagen bundles and associated spindled cells. Myxoid change is common. Since Dr. Enzinger’s description, SCL has been found to express vimentin and CD34, the latter, along with the demographics, being a hallmark of the tumor. But unlike SFT, it is only rarely positive for CD99 or bcl-2. Ultrastructural findings suggest that SCL consists of a biphasic population of adipocytes and undifferentiated mesenchymal cells.
The case we are presenting, by H&E appearance, location of the tumor, patient gender and CD34 positivity, favored an interpretation of SCL. It also showed classic patterns of a SFT including prominent ectatic “hemangiopericytoma-like†vessels with perivascular hyalinization. There was also a distinct interface between the SCL and SFT phenotypes suggesting either a modulation between expressions of the same tumor or an abutment between two different, but morphologically similar processes. The literature, however, describes a case of “spindle cell lipoma like tumor†which had immunohistochemical findings similar to our case and has suggested that at least a subset of spindle cell lipomas may represent forms of SFT.
The cytogenetic findings in the two tumors are disparate. SCL commonly shows aberrations involving 16q and less commonly 13q and 6p while those of SFT include trisomy 21, t(2:3)(p21:p26) and trisomy 8. It has also been pointed out that involvement of subcutaneous tissue of the posterior neck, shoulder and upper back is common for SCL, the location is unusual in SFT.
It is of interest that while recent reports have described a variant of SFT as “lipomatous hemangiopericytoma†(L-HPC), the general current consensus is that the HPC is actually a phenotype and is likely not a true neoplasm. It is felt that the characteristic vascular pattern of “HPC†is also commonplace in SFT and that many adult “HPCs†are actually SFT variants The prominent hyalinized vasculature seen in our case is quite similar to that described as L_SFT/L_HPC, excepting that our vessels were mostly rounded, rather than “staghornâ€. The spectrum of allowable changes in SFT is likely to expand as more entities are felt to be related to SFT.
In summary, this case fulfilled morphologic criteria for both spindle cell lipoma (SCL) and solitary fibrous tumor (SFT). However the immunohistochemical findings of positivity for CD34, CD99, bcl-2 and vimentin suggest a closer relationship to SFT and suggest that there is a spectrum of changes between the two neoplasms. Our case also suggests the possibility that at least some of the cases previously described as L-HPC may in fact represent combined SCL and SFT. Further work needs to be done to delineate this.
Suggested reading:
Cameselle-Teijeiro J; Manuel Lopes J; Villanueva JP; Gil-Gil P; Sobrinho-Simoes M. Lipomatous haemangiopericytoma (adipocytic variant of solitary fibrous tumour) of the thyroid. Histopathology 43(4): p406-8, 2003.
Farah-Klibi F; Ferchichi L; Zairi I; Rammeh S; Adouani A; Jilani SB; Zermani R. Lipomatous hemangiopericytoma (adipocytic variant of solitary fibrous tumor) of the orbit. A case report with review of the literature. Pathologica 98(6): p645-8, 2006.
Gengler C; Guillou L. Solitary fibrous tumour and haemangiopericytoma: evolution of a concept. Histopathology 2006 Jan;48(1): p63-74.
Li Y; Chang Q; Rubin BP; Fletcher CD; Morgan TW; Mentzer SJ; Sugarbaker DJ; Fletcher JA; Xiao S. Hemangiopericytoma: histopathological pattern or clinicopathologic entity? Semin Diagn Pathol 12(3): p221-32, 1995.
Moran CA; Suster S; Koss MN. The spectrum of histologic growth patterns in benign and malignant fibrous tumors of the pleura. Semin Diagn Pathol 9(2): p169-80, 1992.
Nappi O; Ritter JH; Pettinato G; Wick MR. Insulin receptor activation in solitary fibrous tumours. J Pathol 211(5): p550-4, 2007.
Yamaguchi T; Takimoto T; Yamashita T; Kitahara S; Omura M; Ueda Y. Fat-containing variant of solitary fibrous tumor (lipomatous hemangiopericytoma) arising on surface of kidney. Urology 65(1): p175, 2005.
Yamazaki K; Eyden BP. Pulmonary lipomatous hemangiopericytoma: report of a rare tumor and comparison with solitary fibrous tumor. Ultrastruct Pathol 31(1): p51-61, 2007.