 History: A 40-year-old male presented with a 10 day history of abdominal pain, distension and vomiting. The clinical diagnosis of intestinal obstruction resulted in a subsequent hemicolectomy.
History: A 40-year-old male presented with a 10 day history of abdominal pain, distension and vomiting. The clinical diagnosis of intestinal obstruction resulted in a subsequent hemicolectomy.
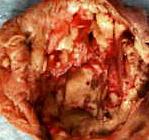
The excised specimen included terminal ileum, colon, cecum and appendix. The colon and cecum were dilated and had thickened walls with spotty whitish mucosal plaques with necrosis (Fig. 1). The specimen had a bad odor even after fixation.
The large bowel contained numerous “flask-like†ulcerations covered by fibrinous exudate separated by normal intervening mucosa (Figs. 1a,2,3). Chronic inflammatory cell infiltrates in the mucosa and submucosa were present as were scattered organisms bearing a resemblance to histiocytes but having a nucleolus-like structure. These trophozoites measured 15-25 microns in diameter, were rounded or ovoid and contained an abundant cytoplasm with a distinctive vacuolation (Fig. 4).
Diagnosis: “Colonic amebiasisâ€
Wafaa Elatre, MD, Fouad Abdelhalim, MD, Donald R. Chase MD
Department of Pathology and Human Anatomy,
Loma Linda University and Medical Center, Loma Linda, CA
California Tumor Tissue Registry, Loma Linda, CA
Discussion: Amebiasis is caused by the protozoan parasite Entamoeba histolytica. Infection is acquired by a fecal-oral route, either by person-to-person contact or indirectly by eating or drinking fecally contaminated food or water. The infestation occurs worldwide, especially in regions with poor sanitation. E. histolytica antibody prevalence rates (reflecting past or recent infection), commonly range from 6% to 25% in developing countries, but may exceed 50% in some communities.
The incubation period is generally 2-4 weeks but ranges from a few days to years. The clinical spectrum of intestinal amebiasis ranges from asymptomatic infection to fulminant colitis and peritonitis. The parasite initially infects the colon, but it occasionally may spread to other organs, most commonly the liver (amebic liver abscess). In persons infected with E. histolytica who are symptomatic, the most common symptom is diarrhea. The diarrhea can result in painful, bloody bowel movements, with or without fever (amebic dysentery).
Asymptomatic human infections are usually diagnosed by finding cysts shed with the stool. Various flotation or sedimentation procedures have been developed to recover the cysts from fecal matter and stains help to visualize the isolated cysts for microscopic examination. Since cysts are not shed constantly, a minimum of 3 stools should be examined. In symptomatic infections, the motile form (the trophozoite) can often be seen in fresh feces. Serological tests exist and most individuals (whether symptomatic or not) will test positive for the presence of antibodies. The levels of antibody are much higher in individuals with liver abscesses. Serology only becomes positive about two weeks after infection. More recent diagnostic developments include a kit that detects the presence of ameba proteins in the feces and another that detects ameba DNA in feces. These tests are not in widespread use due to their expense
Microscopy is still by far the most widespread method of diagnosis around the world, however it is not as sensitive or accurate as are the afore-mentioned tests. It is important to distinguish E. histolytica cysts from the cysts of nonpathogenic intestinal protozoa such as Entamoeba coli. E. histolytica cysts have a maximum of four nuclei, while Entamoeba coli may have up to 8 nuclei. Additionally, in E. histolytica, the endosome is centrally located in the nucleus, while it is off-center in Entamoeba coli. Finally, chromatoidal bodies in E. histolytica are rounded, while they are jagged in Entamoeba coli. However, another species, Entamoeba dispar, is also a commensal and cannot be distinguished from E. histolytica under the microscope. As E. dispar is much more common than E. histolytica in most parts of the world this means that overcalls of E. histolytica infection take place. The WHO recommends that infections diagnosed by microscopy alone should not be treated if they are asymptomatic and there is no other reason to suspect that the infection is actually E. histolytica.
Suggested Readings:
• Ravdin JI, editor. Amebiasis. Human infection by Entamoeba histolytica. New York: John Wiley & Sons; 1988.
• Ravdin JI. Entamoeba histolytica (amebiasis). In: Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas and Bennett’s principles and practice of infectious diseases. 5th ed. Philadelphia, PA: Churchill Livingstone, pp 2798-810, 2000.
• Sanuki JT, Asai E, Okuzawa S, et al. Identification of Entamoeba histolytica and E. dispar cysts in stool by polymerase chain reaction. Parasitol Res, 83:96-8, 1997.
• Tanyuksel M, Petri WA Jr. Laboratory diagnosis of amebiasis. Clin Microbiol Rev, 6:713-29, 2003.
• Verweij JJ, Blotkamp J, Brienen A, et al. Differentiation of Entamoeba histolytica and Entamoeba dispar cysts using polymerase chain reaction on DNA isolated from faeces with spin columns. Eur J Clin Microbiol Infect Dis,19:358-61, 2000.