 History: A 71-year-old male presented with persistent headache. CT and MRI scans revealed a 5.0 x 4.0 x 2.5 cm bifrontal brain parenchymal mass with two smaller dural-based satellite lesions measuring 3.0 and 1.0 cm in greatest diameter (Fig. 1). The masses were contrast-enhancing and preoperatively were interpreted as meningiomas. During surgery, multiple pieces of pink-gray, rubbery tissue fragments with a lobulated appearance were collected which aggregated 15.0 x 14.0 x 2.5 cm.
History: A 71-year-old male presented with persistent headache. CT and MRI scans revealed a 5.0 x 4.0 x 2.5 cm bifrontal brain parenchymal mass with two smaller dural-based satellite lesions measuring 3.0 and 1.0 cm in greatest diameter (Fig. 1). The masses were contrast-enhancing and preoperatively were interpreted as meningiomas. During surgery, multiple pieces of pink-gray, rubbery tissue fragments with a lobulated appearance were collected which aggregated 15.0 x 14.0 x 2.5 cm.
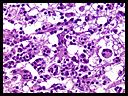
Microscopically the tissue consisted of nodular infiltrates of large histiocytes intermixed with small lymphocytes and plasma cells surrounded and focally compressed by a fibrotic background (Fig. 2,3). Lymphoplasmacytic cuffing of blood vessels was present. No frank necrosis, or granulomata, or eosinophilia were identified. Microorganisms were not detected by special stains.
The pale-staining histiocytes showed indistinct cellular margins with eosinophilic, vacuolated cytoplasm and large eccentric nuclei. The nuclei were rounded to oval with a vesicular chromatin pattern. Occasional histiocytes were multinucleated. The chronic inflammatory infiltrate consisted of histiocytes displaying prominent emperipolesis. The phagocytized cells were primarily intact small lymphocytes and plasma cells within the cytoplasm of the large histiocytes. The histiocytes were diffusely positive for S-100 and CD68, but were negative for CD1a. The tumor was negative for GFAP and AE1-3, but EMA positivity was focally observed. Ki-67 was rarely positive in nuclei, primarily in the inflamed lymphocytic cells and plasmacytoid cells (Fig. 4). No evidence of underlying neuronal or glial neoplasm was identified. Staining for CD20 and CD3 showed a mixed population of B and T lymphocytes in the background. Kappa and lambda light chain immunohistochemistry showed polytypic staining in the plasma cell infiltrates.
Diagnosis: “Rosai-Dorfman Disease of the Central Nervous Systemâ€
Mingyi Chen, MD, and Donald R. Chase, MD
Department of Pathology, Loma Linda University and Medical Center
California Tumor Tissue Registry
Discussion: Sinus histocytosis with massive lymphadenopathy, also known as Rosai-Dorfman Disease (RDD), is an idiopathic histocytic proliferation usually affecting lymph nodes. In 1969, Rosai and Dorfman first described this benign histiocytic proliferative disorder and named it “sinus histiocytosis with massive lymphadenopathyâ€.
The literature reports that about 600 cases of RDD have been described through 2004. The disease occurs in all races but mainly in whites (43%). The classic clinical presentation is painless cervical lymphadenopathy, but it can also present with nasal obstruction, tonsillar enlargement, or hearing abnormalities. The disease occurs most often in the third and fourth decades and may be accompanied by leukocytosis, elevated erythrocyte sedimentation rate (ESR), weight loss, and possibly hypergammaglobulinemia. While lymph node involvement often dominates the clinical presentation, other anatomic sites are involved in 30-40% of cases, most commonly skin, upper respiratory tract, orbit and/or testes. Histologically, the lesions consist of variable numbers of pale-staining histocytes with characteristic emperipolesis often overshadowed by extensive lymphoplasmacytic infiltrates and fibrosis in the background. Special stains for organisms are generally negative. The large histiocytes are positive for S100 protein and CD68, but negative for CD1a. Examinations of immunostained sections can facilitate the recognition of emperipolesis. The engulfed cells are most often lymphocytes, but plasma cell and erythrocytes may also be occasionally seen.
Involvement of the central nervous system (CNS) is particularly uncommon (<5%) and is generally not accompanied by lymphadenopathy. CNS-RDD can present at any age but usually involves patients between 20 and 40 years of age, again with a slight male predominance. Approximately 75% of cases are intracranial whereas 20% involve the spine. Symptoms vary depending on the location of the primary lesion. Symptoms of intracranial disease include seizures, headache, weakness, and cranial nerve palsy. Over 90% of CNS-RDD cases involve the leptomeninges and are seen by neuro-imaging as a dural-based, contrast-enhancing masses that often elicit vasogenic edema in the underlying cerebral cortex and white matter. Thus, clinically and radiologically, the disease is originally thought to represent a meningioma. Definitive diagnosis of RDD involving the leptomeninges is established by biopsy and pathologic examination.
Histopathologically, RDD of the leptomeninges has features similar to that of lymph node disease. Generally the dura is thickened, fibrotic and contains a variable density of chronic inflammatory cells dominated by lymphocytes and plasma cells. Occasionally, neutrophils and eosinophils are noted. Either interspersed or in sheets are large pale histiocytes with abundant vacuolated cytoplasm. Although emperipolesis is particularly characteristic of RDD of the leptomeninges, only about 30% of cases show this feature. In some cases, the non-histocytic infiltrates obscure the histiocytic population. Binucleate plasma cells and intracytoplasmic eosinophilic inclusions (Russell bodies) are commonly observed. Blood vessels are frequently outlined by cuffs of plasma cells and/or lymphocytes. Necrosis, well-formed granulomata, and eosinophilia have been consistently absent. Before defined by Rosai and Dorfman, the entity was usually diagnosed as plasma cell granuloma or inflammatory pseudotumor.
The etiology of RDD is not well understood. Most authors have suggested that it represents an autoimmune disease. Although an infection has been suggested as an underlying cause, a definitive infectious agent has never been discovered. Currently it is considered a benign, idiopathic histiocytosis. The studies of immunophenotypic profile and monokine expression demonstrate that interleukin-I and tumor necrosis factor-alpha are derived from activated macrophages.
Histological differential diagnostic considerations of CNS-Rosai-Dorfman disease include primary CNS lesions, inflammatory pseudotumors, granulomatous diseases such as Wegener’s graulomatosis, and sarcoidosis, Hodgkin disease, Primary T-cell-rich/ histiocyte-rich B-cell lymphoma, lymphomatoid granulomatosis, and Langerhans histiocytosis. The pathologic work-up should begin with close inspection of H&E slides since the morphologic features of large pale histocytes in combination with lymphophagocytosis are strongly suggestive of Rosai-Dorfman disease. Nonetheless, special stains for organisms should be performed for distinguishing RDD from similar-appearing infectious nonspecific reactive conditions such as tuberculosis.
• Some menigiomas may elicit a pronounced chronic inflammatory reaction, and are called lymphoplasmacytic meningioma. They contain conspicuous plasma cell-lymphocytic components which may be confused with the infiltrates of RDD-CNS. Recognition of typical meningioma histology coupled with confirmatory immunostaining for epithelial membrane antigen readily differentiate these two processes.
• Plasma cell granuloma (inflammatory pseudotumor, inflammatory myofibroblastic tumor, and xanthomatous pseudotumor) is an unusual non-neoplastic lesion. Similar to RDD-CNS, it clinically presents as a discrete, dural-based inflammatory mass comprised of a proliferation of inflammatory cells with a predominance of plasma cells and a variable number of lymphocytes, neutrophils, eosinophils, and histiocytes, in a fibrovascular background. These histological features are almost identical to RDD. Some authors even believe that they are related disorders. In fact, a recent report, after reviewing older cases, found that many previously diagnosed CNS-plasma cell granulomas or inflammatory pseudotumors were, in fact, RDD-CNS. Therefore, it is recommended that before rendering a diagnosis of plasma cell granuloma in the CNS, RDD should be excluded through the careful evaluation for emperipolesis and S100 immunostaining.
• Lymphomatoid granulomatosis (LYG) is a rare Epstein-Barr virus–associated systemic angiodestructive lymphoproliferative disease. It is characterized by prominent pulmonary involvement but can also involve nervous system (25%). Histologically, it shows nodular polymorphic lymphoid infiltrate composed of small lymphocytes, plasma cells, and variable numbers of large atypical mononuclear cells. The typical pathological features of angiitis with transmural lymphocytic infiltration and granulomatosis with central necrosis are not seen in RDD-CNS.
• Langerhans histiocytosis often presents as discrete histiocytic nodules attached to the dura. Cytologically, the cells differ from Rosai-Dorfman histiocytes due to conspicuous longitudinal nuclear grooves and lobulation. Eosinophils, often prominent in Langerhans histiocytoses, are not conspicuous in RDD. In addition, Langerhans histiocytes are CD1a-positive, whereas those of Rosai-Dorfman disease are CD1a-negative. Molecular studies using polymorphic regions of the human androgen receptor locus have demonstrated that RDD is a polyclonal disorder, in contrast to the Langerhans cell histiocytosis, which is monoclonal.
• RDD-CNS can mimic some granulomatous diseases involving the central nervous system. A lack of sheet-like necrobiotic type necrosis and necrotizing capillaritis should essentially exclude Wegener’s granulomatosis; the absence of small, well formed granulomas makes sarcoidosis less likely.
• Because of the presence of fibrosis, RDD-CNS may have a distinctly nodular appearance suggestive of the nodular sclerosing variant of Hodgkin disease. Hodgkin disease occurring in the CNS is extremely rare and is typically associated with relapse. Although classic Reed-Sternberg cells are readily distinguished from RDD histiocytes, mononuclear variants, particularly lacunar cells may mimic RDD histiocytes. Classic Reed-Sternberg cells and variants, however, lack emperipolesis and S100 immunoreactivity and are typically positive for CD15 and CD30.
• Non-Hodgkin lymphoma such as T-cell-rich/ histiocyte-rich B-cell lymphoma (H/TCRBCL) has rare CNS involvement. It contains a minority of large neoplastic cells showing diffuse positively for CD20. Careful immunohistological studies to determine monoclonal expansion of B cells are required for diagnosis of non-Hodgkin B cell lymphoma.
• Intracranial involvement by extramedullary plasmacytoma is rare but well documented. The lesions are usually attached to the dura and radiographically resemble meningioma. This differential diagnosis is easily established by demonstrating the immunohistochemical polyclonality of the plasma cells infiltrates of RDD-CNS.
CNS-Rosai-Dorfman disease is considered a benign condition and in most cases surgical resection is the treatment of choice. The number of cases in the literature is small, and disease progression following surgical resection is uncommon. However disease progression and poor outcome seem to occur most often when the patient has bulky, multi-organ disease. In cases of persistent or progressive disease, a number of adjuvant therapies have been administered with variable success, including corticosteroids, vincristine, cytoxan, and radiation.
Suggested Reading:
1. Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy: a newly recognized benign clinicopathological entity. Arch Pathol 1969; 87:63-70.
2. Rosai J, Dorfman RE Sinus histiocytosis with massive lymphadenopathy: a pseudolymphomatous benign disorder. Analysis of 34 cases. Cancer 1972; 30:1174-88.
3. Foucar E, Rosai J, Dorfman R. Sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease): review of the entity. Sem. Diagn. Pathol 1990; 7:19-73.
4. Andriko JA, Morrison A, Colegial CH, et al. Rosai-Dorfman disease isolated to the central nervous system: a report of 11 cases. Mod Pathol 2001;14:172-178.
5. Siadati A, Powell SZ, Shahab I, et al. Intracranial Rosai-Dorfman disease. Arch Pathol Lab Med 2001;125:1115-1116.
6. Wu M, Anderson AE, Kahn LB. A report of intracranial Rosai-Dorfman disease with literature review. Ann Diagn. Pathol. 2001; 5:96-102.
7. Kattner K.A, Stroink AR, Roth TC. et al. Rosai-Dorfman disease mimicking parasagittal meningioma: case presentation and review of the literature. Surg Neurol 2000;53:452-457.
8. Arber DA, Weiss LM, Chang KL. Detection of Epstein-Barr virus in inflammatory pseudotumor. Semin Diagn Pathol 1998;15:155–160.